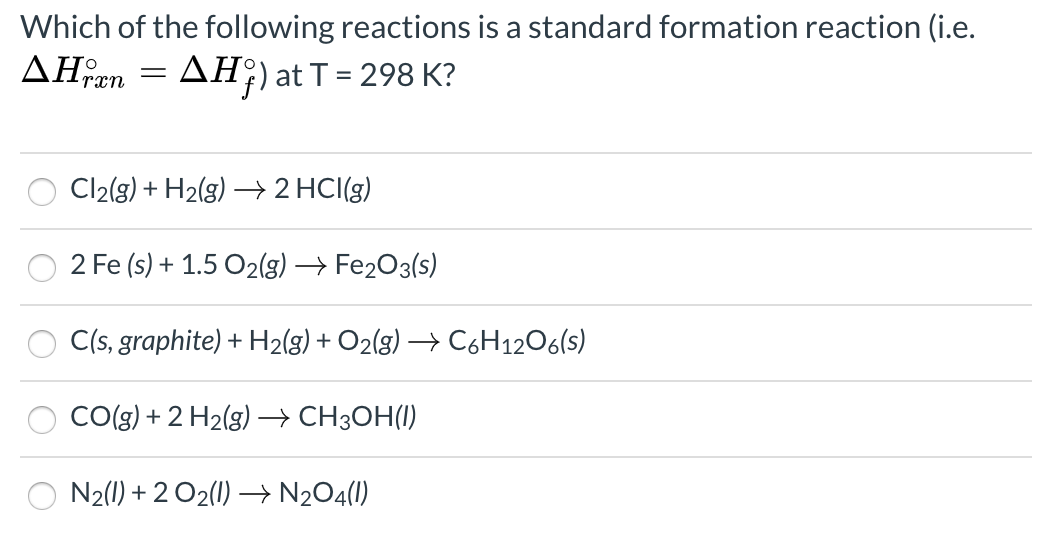Standard Formation Reaction Of Liquid Ethanol - Balanced chemical equation for the formation of ethanol the standard formation reaction of liquid ethanol (c2h5oh) involves the combination of. The standard enthalpy of formation, δh o f, is the enthalpy change for a formation equation when all substances are in their standard states. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. What is the standard enthalpy of formation for ethanol c 2h 5oh? Determine the standard enthalpy of formation for ethylene glycol. Well, this site quotes δh ∘ f (ethanol) = − 277.7 ⋅ kj ⋅ mol−1. 1) the first thing to do is look up standard enthalpies of formation for the.
193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. What is the standard enthalpy of formation for ethanol c 2h 5oh? The standard enthalpy of formation, δh o f, is the enthalpy change for a formation equation when all substances are in their standard states. Well, this site quotes δh ∘ f (ethanol) = − 277.7 ⋅ kj ⋅ mol−1. Determine the standard enthalpy of formation for ethylene glycol. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. Balanced chemical equation for the formation of ethanol the standard formation reaction of liquid ethanol (c2h5oh) involves the combination of. 1) the first thing to do is look up standard enthalpies of formation for the.
1) the first thing to do is look up standard enthalpies of formation for the. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. Determine the standard enthalpy of formation for ethylene glycol. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation, δh o f, is the enthalpy change for a formation equation when all substances are in their standard states. Balanced chemical equation for the formation of ethanol the standard formation reaction of liquid ethanol (c2h5oh) involves the combination of. Well, this site quotes δh ∘ f (ethanol) = − 277.7 ⋅ kj ⋅ mol−1. What is the standard enthalpy of formation for ethanol c 2h 5oh?
Solved Which of the following reactions is a standard
1) the first thing to do is look up standard enthalpies of formation for the. Well, this site quotes δh ∘ f (ethanol) = − 277.7 ⋅ kj ⋅ mol−1. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. The standard enthalpy of formation, δh o f,.
SOLVED a) Write a balanced chemical equation for the standard enthalpy
Determine the standard enthalpy of formation for ethylene glycol. The standard enthalpy of formation, δh o f, is the enthalpy change for a formation equation when all substances are in their standard states. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. Well, this site quotes.
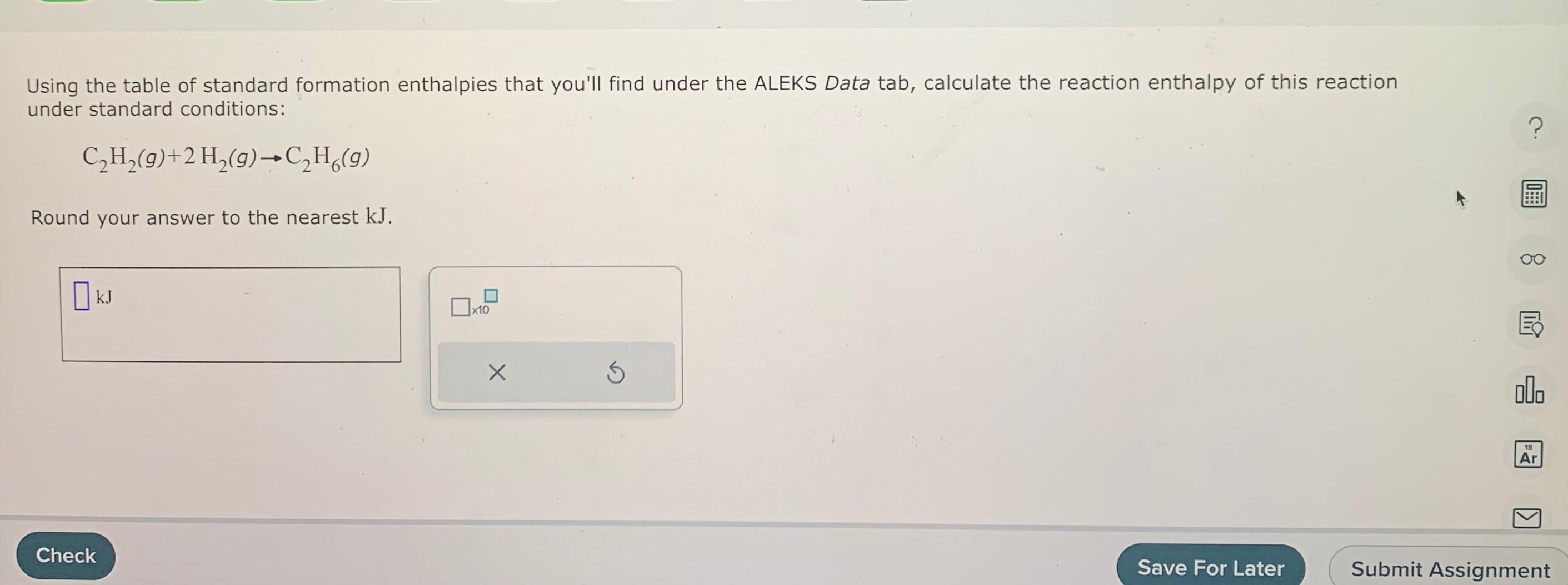
Solved Using the table of standard formation enthalpies that
1) the first thing to do is look up standard enthalpies of formation for the. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. Balanced chemical equation for the formation of ethanol the standard formation reaction of liquid ethanol (c2h5oh) involves the combination of. 193 rows in.
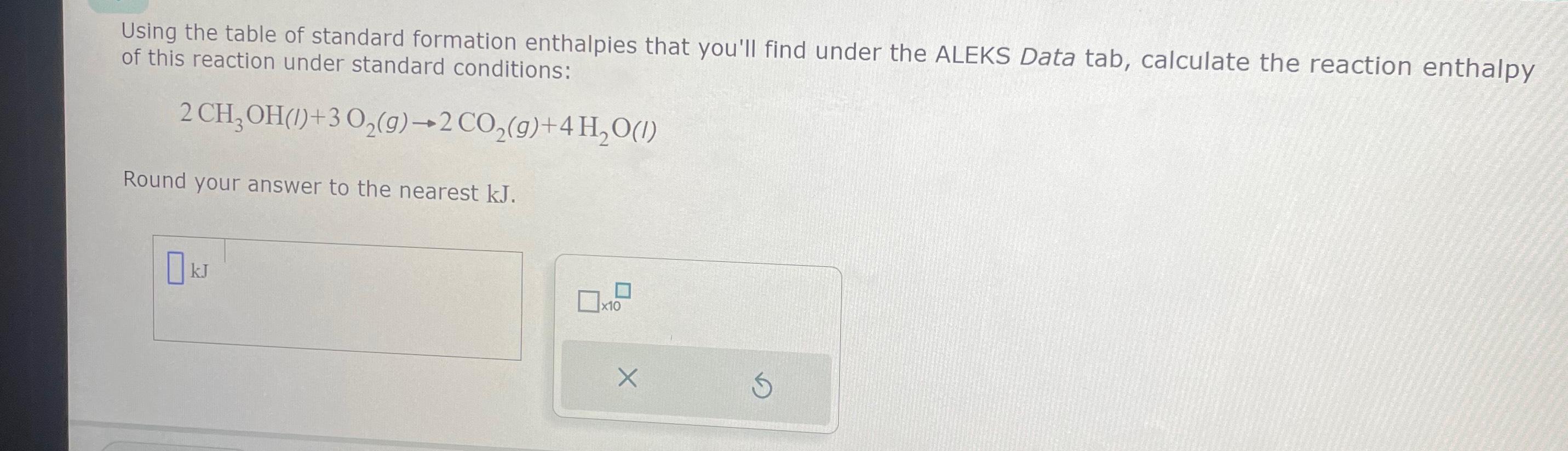
Solved Using the table of standard formation enthalpies that
What is the standard enthalpy of formation for ethanol c 2h 5oh? 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. Determine the standard enthalpy of formation for ethylene glycol. 1) the first thing to do is look up standard enthalpies of formation for the. Balanced.
SOLVED6. Calculate the standard enthalpy change (AH) for the
Balanced chemical equation for the formation of ethanol the standard formation reaction of liquid ethanol (c2h5oh) involves the combination of. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. Determine the standard enthalpy of formation for ethylene glycol. 193 rows in chemistry and thermodynamics, the standard enthalpy.
PPT Heat of Formation PowerPoint Presentation, free download ID3890043
1) the first thing to do is look up standard enthalpies of formation for the. Well, this site quotes δh ∘ f (ethanol) = − 277.7 ⋅ kj ⋅ mol−1. Balanced chemical equation for the formation of ethanol the standard formation reaction of liquid ethanol (c2h5oh) involves the combination of. What is the standard enthalpy of formation for ethanol c.
Vidéo question Déterminer l’enthalpie standard de formation de l
Determine the standard enthalpy of formation for ethylene glycol. What is the standard enthalpy of formation for ethanol c 2h 5oh? 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. 1) the first thing to do is look up standard enthalpies of formation for the. Balanced.
Standard Enthalpy of Formation and Formation Reactions OpenStax
1) the first thing to do is look up standard enthalpies of formation for the. Well, this site quotes δh ∘ f (ethanol) = − 277.7 ⋅ kj ⋅ mol−1. Determine the standard enthalpy of formation for ethylene glycol. Balanced chemical equation for the formation of ethanol the standard formation reaction of liquid ethanol (c2h5oh) involves the combination of. 193.
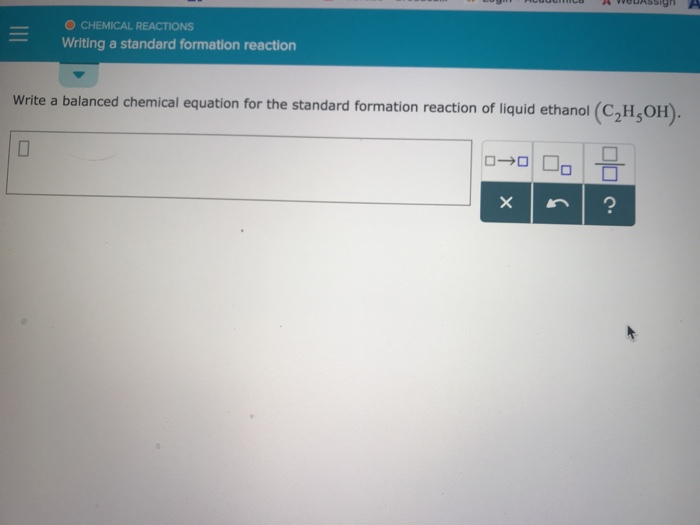
Solved O CHEMICAL REACTIONS Writing a standard formation
What is the standard enthalpy of formation for ethanol c 2h 5oh? 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. The standard enthalpy of formation, δh o f, is the enthalpy change for a formation equation when all substances are in their standard states. 1) the.
SOLVED 'Write a balanced chemical equation for the standard formation
Well, this site quotes δh ∘ f (ethanol) = − 277.7 ⋅ kj ⋅ mol−1. What is the standard enthalpy of formation for ethanol c 2h 5oh? The standard enthalpy of formation, δh o f, is the enthalpy change for a formation equation when all substances are in their standard states. Balanced chemical equation for the formation of ethanol the.
The Standard Enthalpy Of Formation, Δh O F, Is The Enthalpy Change For A Formation Equation When All Substances Are In Their Standard States.
1) the first thing to do is look up standard enthalpies of formation for the. Well, this site quotes δh ∘ f (ethanol) = − 277.7 ⋅ kj ⋅ mol−1. Balanced chemical equation for the formation of ethanol the standard formation reaction of liquid ethanol (c2h5oh) involves the combination of. What is the standard enthalpy of formation for ethanol c 2h 5oh?
136 Rows Standard Enthalpy Change Of Formation (Data Table) These Tables Include Heat Of Formation Data Gathered From A Variety Of Sources,.
193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. Determine the standard enthalpy of formation for ethylene glycol.