Vaccine Temperature Excursion Form - Use this form to document any unacceptable vaccine storage event, such as exposure of vaccines to temperatures that are outside the. It saves the cost of replacing. This form should be used to document any temperature excursions that your site experiences. • if vaccines were moved to another unit please provide 3 days stable temps in range before moving vaccines back into unit. Keep detailed notes of all. The best way to protect vaccine quality is to follow proper vaccine storage and handling specifications. Use this form to document any unacceptable vaccine storage event, such as exposure of refrigerated vaccines to temperatures that are.
The best way to protect vaccine quality is to follow proper vaccine storage and handling specifications. Keep detailed notes of all. Use this form to document any unacceptable vaccine storage event, such as exposure of vaccines to temperatures that are outside the. Use this form to document any unacceptable vaccine storage event, such as exposure of refrigerated vaccines to temperatures that are. It saves the cost of replacing. • if vaccines were moved to another unit please provide 3 days stable temps in range before moving vaccines back into unit. This form should be used to document any temperature excursions that your site experiences.
• if vaccines were moved to another unit please provide 3 days stable temps in range before moving vaccines back into unit. This form should be used to document any temperature excursions that your site experiences. Use this form to document any unacceptable vaccine storage event, such as exposure of refrigerated vaccines to temperatures that are. The best way to protect vaccine quality is to follow proper vaccine storage and handling specifications. Keep detailed notes of all. It saves the cost of replacing. Use this form to document any unacceptable vaccine storage event, such as exposure of vaccines to temperatures that are outside the.
Tips for Maintain Proper Flu Vaccine Storage Temperature
Use this form to document any unacceptable vaccine storage event, such as exposure of vaccines to temperatures that are outside the. Use this form to document any unacceptable vaccine storage event, such as exposure of refrigerated vaccines to temperatures that are. This form should be used to document any temperature excursions that your site experiences. • if vaccines were moved.
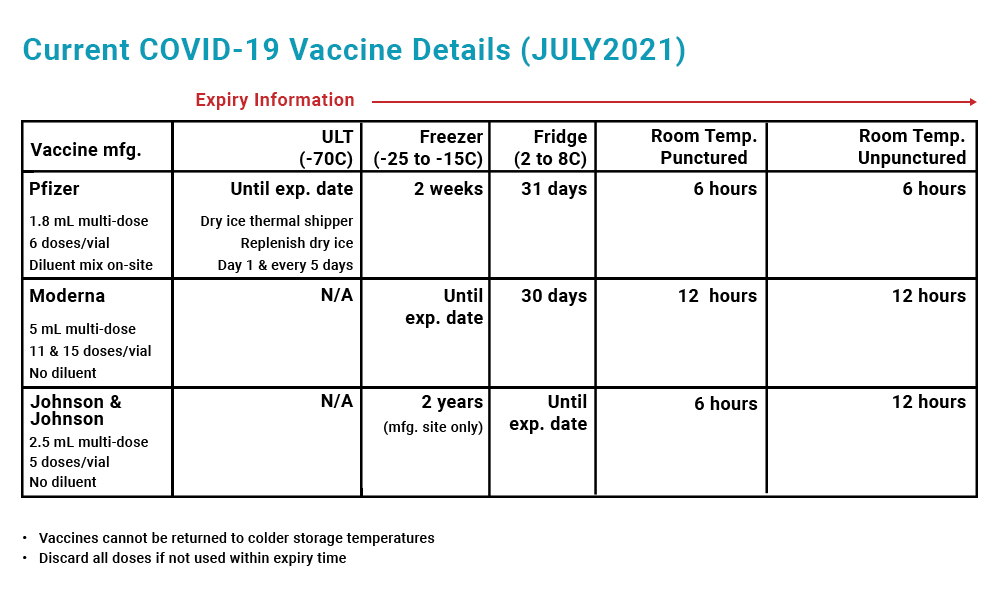
COVID19 Vaccine Temperature Stability Chart TruMed Systems
Use this form to document any unacceptable vaccine storage event, such as exposure of refrigerated vaccines to temperatures that are. Use this form to document any unacceptable vaccine storage event, such as exposure of vaccines to temperatures that are outside the. The best way to protect vaccine quality is to follow proper vaccine storage and handling specifications. Keep detailed notes.
Temperature log for refrigerator fahrenheit Fill out & sign online
This form should be used to document any temperature excursions that your site experiences. Use this form to document any unacceptable vaccine storage event, such as exposure of vaccines to temperatures that are outside the. • if vaccines were moved to another unit please provide 3 days stable temps in range before moving vaccines back into unit. Keep detailed notes.
Coronavirus Vaccine The optimal temperature for storage of front
Keep detailed notes of all. This form should be used to document any temperature excursions that your site experiences. It saves the cost of replacing. The best way to protect vaccine quality is to follow proper vaccine storage and handling specifications. Use this form to document any unacceptable vaccine storage event, such as exposure of vaccines to temperatures that are.
Shingles Vaccine RxDx Healthcare
• if vaccines were moved to another unit please provide 3 days stable temps in range before moving vaccines back into unit. It saves the cost of replacing. This form should be used to document any temperature excursions that your site experiences. The best way to protect vaccine quality is to follow proper vaccine storage and handling specifications. Use this.
Vaccine Cold Storage Rooms How They Work and are Maintained
Use this form to document any unacceptable vaccine storage event, such as exposure of refrigerated vaccines to temperatures that are. This form should be used to document any temperature excursions that your site experiences. • if vaccines were moved to another unit please provide 3 days stable temps in range before moving vaccines back into unit. Use this form to.
Temperature Monitoring and Excursions
Use this form to document any unacceptable vaccine storage event, such as exposure of vaccines to temperatures that are outside the. • if vaccines were moved to another unit please provide 3 days stable temps in range before moving vaccines back into unit. The best way to protect vaccine quality is to follow proper vaccine storage and handling specifications. Use.
Vaccine Temperature Monitoring Best Practices Dickson
Use this form to document any unacceptable vaccine storage event, such as exposure of vaccines to temperatures that are outside the. Use this form to document any unacceptable vaccine storage event, such as exposure of refrigerated vaccines to temperatures that are. This form should be used to document any temperature excursions that your site experiences. It saves the cost of.
The Price of Prevention Vaccine Costs Are Soaring The New York Times
The best way to protect vaccine quality is to follow proper vaccine storage and handling specifications. This form should be used to document any temperature excursions that your site experiences. • if vaccines were moved to another unit please provide 3 days stable temps in range before moving vaccines back into unit. Use this form to document any unacceptable vaccine.
COVID19 Vaccine Temperature Stability Chart TruMed Systems
Use this form to document any unacceptable vaccine storage event, such as exposure of vaccines to temperatures that are outside the. • if vaccines were moved to another unit please provide 3 days stable temps in range before moving vaccines back into unit. It saves the cost of replacing. Keep detailed notes of all. Use this form to document any.
It Saves The Cost Of Replacing.
Use this form to document any unacceptable vaccine storage event, such as exposure of vaccines to temperatures that are outside the. This form should be used to document any temperature excursions that your site experiences. • if vaccines were moved to another unit please provide 3 days stable temps in range before moving vaccines back into unit. The best way to protect vaccine quality is to follow proper vaccine storage and handling specifications.
Keep Detailed Notes Of All.
Use this form to document any unacceptable vaccine storage event, such as exposure of refrigerated vaccines to temperatures that are.